What you should know about the COVID-19 vaccines
January 20, 2021
In December 2020, the Food and Drug Administration (FDA) approved two vaccines recommended to prevent coronavirus (COVID-19), the Pfizer-BioNTech vaccine, and Moderna’s vaccine. Entering the new year with the pandemic still prevalent in everyday life, students and community members alike can inform themselves on both vaccines here.
The Centers for Disease Control and Prevention (CDC) says the vaccines help individuals develop the immunity they need against the virus without developing the illness. They are mRNA vaccines, meaning they don’t use live forms of the virus in the vaccine, they use a harmless piece of the “spike protein,” in the virus therefore there is no risk of contracting COVID-19 from them. The CDC labels them as the “new approach to vaccines,” prompting an individual’s body to develop the necessary protein found on the surface of the virus, and allowing the body to build its own immunity to the virus with it.
Both of the approved vaccines involve two sets of shots, to be administered the second time three to four weeks after the first. The most common side effects include pain, swelling or redness in the area where you get the shot, and chills, tiredness or headaches (which are more likely after the second dose of the vaccine), per the CDC.
According to Harvard Medical School’s Coronavirus Resource Center, the Moderna vaccine has a 94% success rate in preventing COVID-19, while the Pfizer vaccine reportedly reduced the risk of COVID-19 by 95% – according to results published in the New England Journal of Medicine.
Natalie Swisher, University of New Hampshire senior and biomedical science major, is an EMT at McGregor EMS, the 911 ambulance provider that covers UNH, Durham, Lee and Madbury. She received the Moderna vaccine on Jan. 5 and said in an email that it went “incredibly smooth.” Swisher got the vaccine at Exeter High School, waited in her car the entire time, and was set to leave after 15 minutes. She noted some initial arm soreness but no other symptoms about ten hours after administration.
“I would 110 percent recommend anyone and everyone get the vaccine if they’re able to do so. Any steps toward a more normal future are ones I’m willing to take, especially ones that make working on the ambulance safer for myself as well as patients,” Swisher said.
She registered through the Vaccine Administration Management System (VAMS) online through the CDC. Moderna was chosen for her when she arrived – it is the easier of the two vaccines to transport, per CNN, because it can be moved in a normal freezer rather than a “super cold transportation network.” Swisher said the arm pain faded completely after two days, and she’s had no other symptoms. She was able to go about her day like normal afterwards, she said.
Stephen Mackenzie, 63, got the Pzfizer vaccine because it was the first one available to him. He reported the same arm soreness as Swisher, and noted he’d expect the other side effects (chills, tiredness etc.) after his second administration, if he were to experience them at all.
Mackenzie said he was “anxious to get it,” and jumped on the opportunity as he’s a Medication Nursing Assistant at Saint Ann Rehabilitation and Nursing Center in Dover, New Hampshire, but noted it was unfortunate that his wife wasn’t able to yet.
The CDC encourages individuals to get the vaccine even if they have already had COVID-19 and recovered, considering it is unknown how long natural immunity (from being infected) of COVID-19 lasts. Harvard’s Coronavirus Resource Center says the vaccines can prevent you from becoming sick, but it may not prevent you from infecting others.
According to The New York Times vaccine tracker, 20 more vaccines are in “phase three” large-scale efficacy tests as of Jan. 14.
The CDC records 12,279,180 people in the United States have received the first dose of a COVID-19 disease with over 31 million doses distributed as of Jan. 18. The CDC is using v-safe, a symptom tracking smartphone tool, to allow patients who received the vaccine to document their symptoms for the CDC’s records.
NHPR (New Hampshire Public Radio) reported in its COVID-19 tracker that over 86,000 doses of the vaccines have been delivered to vaccination sites in New Hampshire, with 60,761 doses currently administered.
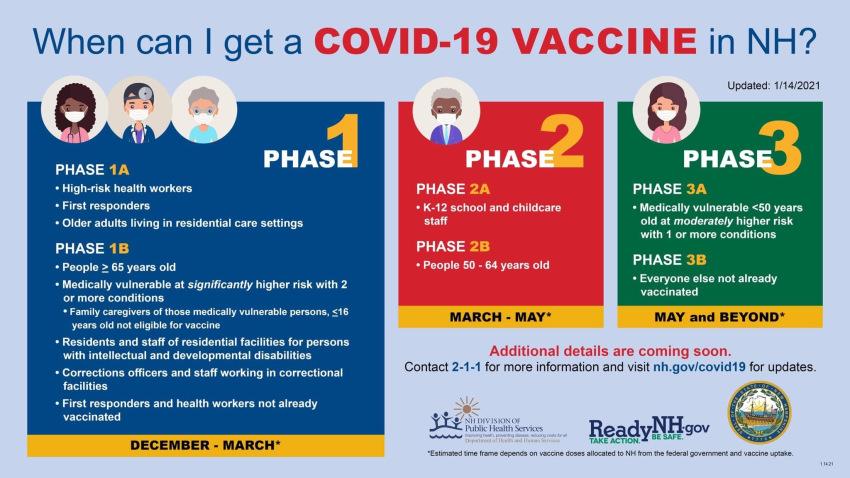
Photo Courtesy of NH.gov